The higher the energy-level number, the farther away the electron is from the nucleus - and the higher the energy.īohr also found that the various energy levels can hold differing numbers of electrons: energy level 1 may hold up to 2 electrons, energy level 2 may hold up to 8 electrons, and so on. So Bohr numbered the electron’s energy levels. Slight variations in the amount of the energy are seen as light of different colors.īohr found that the closer an electron is to the nucleus, the less energy it needs, but the farther away it is, the more energy it needs. Sometimes the energy released by electrons occupies the portion of the electromagnetic spectrum (the range of wavelengths of energy) that humans detect as visible light.After it’s done being excited, the electron can return to its original ground state by releasing the energy it has absorbed, as shown in the diagram below.This higher-energy, less-stable state is called the electron’s excited state. But it can move to a higher-energy, less-stable level, or shell, by absorbing energy. The energy level an electron normally occupies is called its ground state.He said that the energy of an electron is quantized, meaning electrons can have one energy level or another but nothing in between. Bohr used the term energy levels (or shells) to describe these orbits of differing energy.The Bohr model shows that the electrons in atoms are in orbits of differing energy around the nucleus (think of planets orbiting around the sun).Niels Bohr, a Danish scientist, explained this line spectrum while developing a model for the atom: Bohr said that the electron had to release energy to change its energy so the differences between the energies of light seen in the atomic spectrum should correspond to the differences in energies of the energy levels. He used these energies to find the energies that the single electron in the hydrogen atom could have. Bohr found the energy of the colors of light that the hydrogen atom released. This led Bohr to theorize that electrons only have certain energies in an atom and they had to be in energy levels. Each color corresponds to a specific amount of energy however when the light given off by the hydrogen atom was passed through a prism, only certain colors of light could be seen. When white light is diffracted with a prism, all the colors of the visible spectrum can be seen. The Bohr model was based on his observations of the atomic emissions spectrum of the hydrogen atom. The Rutherford model had several flaws that the Bohr model overcame. 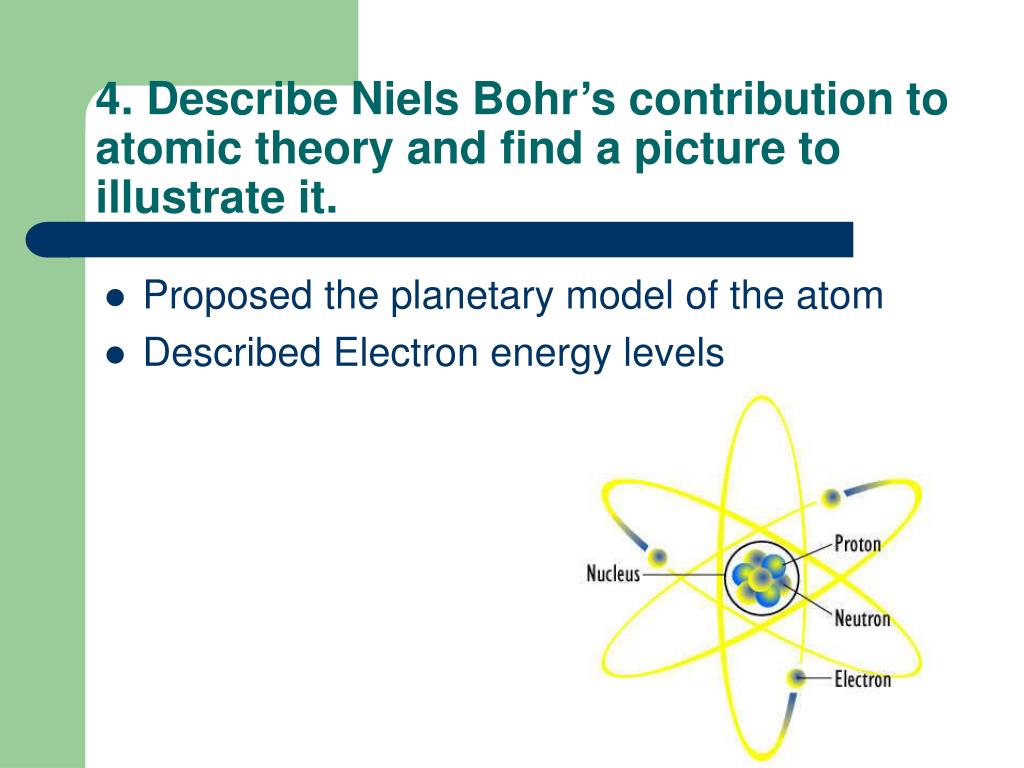
This model was proposed in 1913 by Niels Bohr and was really an expansion on the Rutherford model of 1911. 
The energy difference between the initial and final orbit is emitted by the atom in bundles of electromagnetic radiation called photons. When an electron changes orbits, it does so in a sudden quantum leap. A simple definition of Bohr’s atomic model is: electrons orbit the nucleus at set distances.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
